
Manufacturing Facilities
All our antiviral, antibacterial, and recombinant vaccines are manufactured at our world-class facility in Genome Valley, Hyderabad, India. This state-of-the-art plant is one of the largest of its kind in the Asia-Pacific region and meets the standards set by global regulators such as the USFDA, Saudi Food and Drug Authority (SFDA), South Korea’s Ministry of Food and Drug Safety (formerly KFDA), the UK’s Medicines and Healthcare products Regulatory Agency (MHRA), and the World Health Organization (WHO). Our facility also features one of the finest bacteriology and virology laboratories in the country.
We operate one of the finest bacteriology and virology laboratories in the country. Our manufacturing facilities meet the standards of leading global regulators, including SFDA, MHRA (UK), MFDS (South Korea), NRAs, and WHO.
Our rigorous quality control processes ensure that we consistently deliver safe and effective products trusted by physicians worldwide. Committed to manufacturing excellence, we optimize the use of raw materials and natural resources while minimizing waste wherever possible. We also practice responsible waste management to reduce our environmental impact. Our manufacturing records are comprehensive and readily accessible to regulators during inspections. We maintain a zero-tolerance policy toward data integrity issues.
Besides quality, we seek to achieve industry-leading efficiency to deliver on our promise of affordable vaccines. With our patented HIMAX technology, we played a vital role in bringing down the cost of producing Hepatitis–B vaccines, thereby making it affordable for millions across the country and other developing nations. Some of our facilities include:
- rDNA facility (separate blocks for vaccines and bio-therapeutics).
- Bacterial facility.
- Cell Culture facility.
- Polysaccharide facility.
- Probiotic facility.
- Formulation & Filling facilities.
- Centralised, fully automatic inspection, labeling and packing area.
- Prefabricated, independent GMP modules with HVAC for rDNA, viral and bacterial products.
- P IV Isolation to handle bio-hazardous organisms.
- Analytical & Instrumentation laboratory.
Partnerships
Science advances when there is co-operation. Central to Jolly’s research approach is a collaborative attitude to engaging with world-class research institutions on shared priorities. We partner with several leading Indian and global research organisations to advance science that improves public health.
Research partnerships
- All India Institute of Medical Sciences (AIIMS), New Delhi, India.
- Stanford University, School of Medicine, US.
- Center for Vaccine Development, University of Maryland, US.
- National Institute of Allergy and Infectious Diseases, Washington DC, US
- National Institute of Health (NIH), Washington DC, US.
- Centres for Disease Control & Prevention (CDC), Atlanta, US.
- Department of Biotechnology (DBT), Government of India.
- Department of Science & Technology (DST), Government of India.
- Indian Institute of Science, Bangalore (IISc), India.
- Institute of Genomics and Integrative Biology (IGIB), New Delhi, India.
- Jawaharlal Nehru Centre for Advanced Scientific Research, Bangalore, India.
- National Institute of Virology (NIV), Pune, India.
Exports
International Business Development is a wide area network at Jolly Healthcare with the primary goal of supplying affordable vaccines to the world.
This is achieved through fostering
- Valuable partnerships
- Our products advocacy
- Attainment of product market authorization
- Fulfilling supplies to the public and private markets of the respected country.
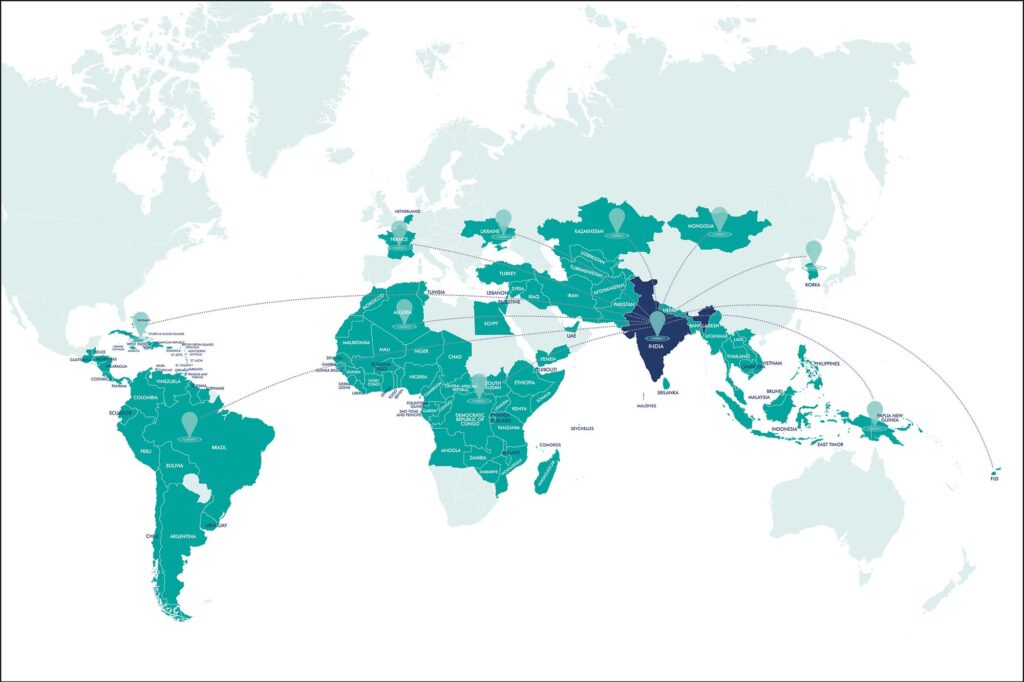
We have been at the forefront in delivering >4 billion doses of vaccines to 123 countries.
JollyRX-B2B Marketplace
At Jolly, we seek to lead innovation in biotechnology in order to lead the fight against disease. We have a rich history of developing breakthrough vaccines and bio-therapeutics to protect people against diseases. Our primary focus is on the neglected region-specific diseases of the developing world. Over 114 multi-disciplinary scientists—the best and the brightest in their respective areas—work collaboratively at our research and development (R&D) facilities.
Jolly has long been known for its world-class research capabilities. We have developed several breakthrough vaccines and bio-therapeutics including the world’s first typhoid conjugate vaccine and recombinant Hepatitis B vaccine.
We own over 145 global patents. Some of our innovative vaccines and therapeutics include Typbar TCV—the world’s first Typhoid Conjugate Vaccine, REGEN-D®—a growth factor gel to treat diabetic foot ulcers developed using recombinant technology, and HIMAX®—a patented technology, which enabled the production of Revac-B mcf®. This first-ever recombinant Hepatitis B vaccine is known for driving highest levels of antigenic recovery compared to other vaccines in its class. We do cutting-edge research in the areas of bioinformatics, molecular epidemiology, molecular biology, microbiology, bacteriology, virology, immunology, biochemistry, clinical research, industrial biotechnology, formulations and product development. We seek to generate and develop path-breaking ideas to target challenging diseases that affect millions of people in the developing world. As a research-led company, we believe in practising the highest standards of integrity. We bring scientific rigour to all stages of product development from discovery and development to clinical trials and manufacturing. With complete adherence to IPR, we work collaboratively with academic and research institutions to undertake fundamental research that helps us address unmet conditions and improve public health.
